|
|
|
| Reliability of nephrolithometric nomograms in patients treated with minimally invasive percutaneous nephrolithotomy: A precision study |
a Department of Urology, San Bassiano Hospital, Bassano del Grappa, Italy
b Department of Urology, University of Trieste, Trieste, Italy
c Institute of Urology, University College Hospitals London, London, UK
d Department of Urology, Minimally Invasive Centre, the first affiliated hospital of Guangzhou Medical University, Guangzhou, China |
|
|
|
|
Abstract Objectives: The study aimed to evaluate quality of nephrolithometric nomograms to predict stone-free rates (SFRs) and complication rates (CRs) in case of minimally invasive percutaneous nephrolithotomy (PNL). In the last decade, nomograms have been introduced to estimate the SFRs and CRs of PNL. However, no data are available regarding their reliability in case of utilization of miniaturized devices. Herein we present a prospective multicentric study to evaluate reliability of Guy’s stone score (GSS), the stone size, tract length, obstruction, number of involved calyces, and essence of stone (S.T.O.N.E.) nephrolithometry score and Clinical Research Office of the Endourological Society (CROES) score in patients treated with minimally invasive PNL. Methods:We evaluated SFRs and CRs of 222 adult patients treated with miniaturized PNL. Patients were considered stone-free if no residual fragments of any size at post-operative unenhanced computed tomography scan. Patients demographics, SFRs, and CRs were reported and analyzed. Performances of nomograms were evaluated with the area under the curve (AUC). Results:We included 222 patients, the AUCs of GSS, CROES score, and S.T.O.N.E. nephrolithometry score were 0.69 (95% confidence interval [CI] 0.61-0.78), 0.64 (95% CI 0.56-0.73), and 0.62 (95% CI 0.52-0.71), respectively. Regarding SFRs, at multivariate binomial logistic regression, only the GSS had significance with an odds ratio of 0.53 (95% CI 0.31-0.95, p=0.04). We did not find significant correlation with complications, with only a trend for GSS. Conclusion:This is the first study evaluating nomograms in miniaturized PNL. They still show good reliability; however, our data showed lower performances compared to standard PNL. We emphasize the need of further studies to confirm this trend. A dedicated nomogram for minimally invasive PNL may be necessary.
|
|
Received: 10 August 2021
Available online: 20 January 2023
|
|
|

| Type of nomogram | Variable evaluated | Nomogram’s subgroup | | GSS [12] | -
Grade 1: solitary stone in the mid- or lower pole; pelvis with normal anatomy | -
Grade 1 | -
Grade 2: solitary stone in upper pole; multiple stones with simple anatomy; any solitary stone with abnormal anatomy | -
Grade 2 | -
Grade 3: multiple stones with abnormal anatomy; stone in diverticulum; partial staghorn | -
Grade 3 | -
Grade 4: staghorn stone; any stone in patient with spina bifida or spinal injury | -
Grade 4 | | S.T.O.N.E. nephrolithometry score [13] | -
Stone size | -
Low complexity [5,6] | -
Tract length | -
Medium complexity [7,8] | -
Presence of obstruction | -
High complexity (>9) | -
Number of calices involved | | -
Mean stone density (essence) | | | CROES score [14] | -
Stone volume | -
Grade 1 (0-100) | -
Stone location | -
Grade 2 (101-150) | -
Prior treatments | -
Grade 3 (151-200) | -
Presence of staghorn | -
Grade 4 (201-350) | -
Number of stones | | -
Case volume per year of the center | |
|
|
Characteristics of GSS, S.T.O.N.E. nephrolithometry score, and CROES score.
|

| Variable | Total | Stone-free case | Residual stone | p-Value | | Patienta | 222 (100) | 173 (77.9) | 49 (22.1) | NA | | Ageb, year | 54 (42-63) | 53 (42-63) | 55 (47-74) | 0.47 | | Gendera | | | | 0.33 | | Female | 93 (41.9) | 69 (39.9) | 24 (49.0) | | | Male | 129 (58.1) | 104 (60.1) | 25 (51.0) | | | Sidea | | | | 0.45 | | Right | 108 (48.6) | 87 (50.3) | 21 (42.9) | | | Left | 114 (51.4) | 86 (49.7) | 28 (57.1) | | | ASA scorea | | | | 0.367 | | 1 | 98 (44.1) | 78 (45.1) | 20 (40.8) | | | 2 | 111 (50.0) | 87 (50.3) | 24 (49.0) | | | 3 | 13 (5.9) | 8 (4.6) | 5 (10.2) | | | 4 | 0 (0.0) | 0 (0.0) | 0 (0.0) | | | Stone burdenb, mm2 | 157 (61-372) | 125 (48-337) | 324 (153-588) | <0.001 | | Stone densityb, HU | 1032 (745-1231) | 1011 (727-1217) | 1093 (771-1276) | 0.18 | | Tract lengthb, mm | 85 (71-100) | 85 (72-102) | 83 (69-96) | 0.16 | | Renal pelvic obstructiona | | | | 0.22 | | None or mild | 160 (72.1) | 126 (72.8) | 34 (69.4) | | | Moderate or severe | 62 (27.9) | 47 (27.2) | 15 (30.6) | | | Calyxes involveda,c | | | | 0.16 | | 1 | 125 (56.3) | 102 (59.0) | 23 (46.9) | | | 2 | 44 (19.8) | 34 (19.7) | 10 (20.4) | | | 3 | 24 (10.8) | 15 (8.7) | 9 (18.4) | | | Multiple (staghorn) | 23 (10.4) | 16 (9.2) | 7 (14.3) | | | Stone locationa,d | | | | 0.03 | | Pelvis | 45 (20.3) | 40 (23.1) | 5 (10.2) | | | Lower calyx | 59 (26.6) | 50 (28.9) | 9 (18.4) | | | Medium calyx | 15 (6.8) | 10 (5.8) | 5 (10.2) | | | Upper calyx | 15 (6.8) | 13 (7.5) | 2 (4.1) | | | Multiple sites | 86 (38.7) | 58 (33.5) | 28 (57.1) | | | Stonea | | | | 0.004 | | Single | 87 (39.2) | 77 (44.5) | 10 (20.4) | | | Multiple | 135 (60.8) | 96 (55.5) | 39 (79.6) | | | Presence of staghorn stonea | | | | <0.001 | | Yes | 54 (24.3) | 31 (17.9) | 23 (46.9) | | | No | 168 (75.7) | 142 (82.1) | 26 (53.1) | | | Prior treatmenta | | | | 0.34 | | None | 181 (81.5) | 137 (79.2) | 44 (89.8) | | | PNL | 20 (9.0) | 16 (9.2) | 4 (8.2) | | | ESWL | 6 (2.7) | 6 (3.5) | 0 (0.0) | | | Endoscopic | 5 (2.3) | 5 (2.9) | 0 (0.0) | | | Multiple | 10 (4.5) | 9 (5.2) | 1 (2.0) | | | S.T.O.N.E. nephrolithometry scoreb | 6 (6-7) | 6 (6-7) | 7 (6-9) | <0.001 | | Guy’s stone scoreb | 2 (1-3) | 2 (1-2) | 3 (2-3) | <0.001 | | CROES nomogramb | 211 (156-269) | 218 (160-270) | 169 (131-230) | 0.004 | | CROES gradeb | 4 (3-4) | 4 (3-4) | 3 (2-4) | <0.001 |
|
|
Demographic and pre-operative data of 222 patients who have undergone minimally invasive PNL.
|

| Nephrolithometric nomogram | Patienta, n (%) | SFR | 30-day CR | | n (%) | p-Value | n (%) | p-Value | | S.T.O.N.E. nephrolithometry score | | | 0.005 | | 0.59 | | 5-6 | 125 (56.3) | 102 (81.6) | | 17 (13.6) | | | 7-8 | 66 (29.7) | 53 (80.3) | | 19 (28.8) | | | 9-13 | 28 (12.6) | 15 (53.6) | | 10 (35.7) | | | GSS | | | <0.001 | | 0.04 | | Grade 1 | 72 (32.4) | 67 (93.1) | | 9 (12.5) | | | Grade 2 | 88 (39.6) | 73 (83.0) | | 13 (14.8) | | | Grade 3 | 38 (17.1) | 21 (55.3) | | 14 (36.8) | | | Grade 4 | 21 (9.5) | 12 (57.1) | | 10 (47.6) | | | CROES system | | | 0.007 | | 0.02 | | Grade 1 (0-100) | 15 (6.8) | 8 (53.3) | | 6 (40.0) | | | Grade 2 (101-150) | 34 (15.3) | 23 (67.6) | | 16 (47.1) | | | Grade 3 (151-200) | 52 (23.4) | 38 (73.1) | | 11 (21.2) | | | Grade 4 (201-350) | 118 (53.2) | 101 (85.6) | | 13 (11.0) | |
|
|
GSS, S.T.O.N.E. nephrolithometry score and CROES score association with SFRs and 30-day CRs.
|

| Variable | Total | Stone-free case | Residual stone | p-Value | | Patienta | 222 | 173 (77.9) | 49 (22.1) | NA | | Maximum tract diameterb, Fr | 16 (14-18) | 16 (14-18) | 18 (14-18) | 0.002 | | Maximum tract diametera, Fr | | | | 0.003 | | 14 | 85 (38.3) | 72 (41.6) | 13 (26.5) | | 16 | 42 (18.9) | 37 (21.4) | 5 (10.2) | | 18 | 94 (42.3) | 64 (37.0) | 30 (61.2) | | 22 | 1 (0.5) | 0 (0.0) | 1 (2.0) | | Puncturea | | | | 0.14 | | 1 | 199 (89.6) | 157 (90.8) | 42 (85.7) | | 2 | 22 (9.9) | 16 (9.2) | 6 (12.2) | | 3 | 1 (0.5) | 0 (0.0) | 1 (2.0) | | Active suctiona | | | | 0.9 | | Yes | 196 (88.3) | 152 (87.9) | 44 (89.8) | | No | 26 (11.7) | 21 (12.1) | 5 (10.2) | | Level of upper puncturea,c | | | | 0.03 | | Below 12th | 134 (60.4) | 112 (64.7) | 22 (44.9) | | Below 11th | 75 (33.8) | 52 (30.1) | 23 (46.9) | | Below 10th | 8 (3.6) | 5 (2.9) | 3 (6.1) | | Patient’s positiona | | | | 0.16 | | Prone | 142 (64.0) | 106 (61.3) | 36 (73.5) | | Supine | 80 (36.0) | 67 (38.7) | 13 (26.5) | | Stone biochemistrya,d | | | | 0.8 | | Calcium oxalate | 150 (67.6) | 116 (67.1) | 34 (69.4) | | Calcium phosphate | 45 (20.3) | 36 (20.8) | 9 (18.4) | | Urates | 9 (4.1) | 8 (4.6) | 1 (2.0) | | Struvite | 10 (4.5) | 7 (4.0) | 3 (6.1) | | Cystine | 5 (2.3) | 3 (1.7) | 2 (4.1) | | Drug-related stone | 1 (0.5) | 1 (0.6) | 0 (0.0) | | Type of drainagea | | | | 0.02 | | Stent plus nephrostomy | 101 (45.5) | 72 (41.6) | 29 (59.2) | | Tubeless | 70 (31.5) | 54 (31.2) | 16 (32.7) | | Totally tubeless | 48 (21.6) | 44 (25.4) | 4 (8.2) | | Length of stayb, day | 3 (2-4) | 3 (1-4) | 4 (3-5) | 0.03 | | 30-day complicationa,d | | | | 0.83 | | Clavien I | 34 (15.3) | 21 (12.1) | 13 (26.5) | | Clavien II | 4 (1.8) | 4 (2.3) | 0 (0.0) | | Clavien IIIa | 3 (1.4) | 2 (1.2) | 1 (2.0) | | Clavien IIIb | 1 (0.5) | 1 (0.6) | 0 (0.0) | | Clavien IVa | 4 (1.8) | 3 (1.7) | 1 (2.0) |
|
|
Peri-operative data of 222 patients who have undergone minimally invasive percutaneous nephrolithotomy included in the study.
|

| Modified Clavien-Dindo | n (%) | | Grade I | 34 (15.3) | | Pain requiring higher analgesia | 21 (9.5) | | Fever | 9 (4.1) | | Diarrhea | 1 (0.5) | | Self-resolving dyspnea | 1 (0.5) | | Clot retention requiring prolonged catheterization | 1 (0.5) | | Urinoma | 1 (0.5) | | Grade II | 4 (1.8) | | Fever requiring antibiotic therapy change | 4 (1.8) | | Grade IIIa | 3 (1.4) | | Hydrothorax | 1 (0.5) | | Displaced stent requiring repositioning under general anesthesia | 1 (0.5) | | Sepsis without organ failure requiring supportive therapy | 1 (0.5) | | Grade IIIb | 1 (0.5) | | Angio-embolization | 1 (0.5) | | Grade IVa | 4 (1.8) | | Sepsis requiring ICU stay | 3 (1.4) | | Pulmonary embolism requiring ICU stay | 1 (0.5) |
|
|
Complications occurred in our study group (n=222) according to Clavien-Dindo classification modified for PNL.
|

| Variable | Univariate | Multivariate | | OR (95% CI) | p-Value | OR (95% CI) | p-Value | | Agea, years | 0.99 (0.97-1.01) | 0.48 | | | | Sex | | Male | 1.00 (Ref.) | | | | | Female | 0.69 (0.36-1.31) | 0.26 | | | | Side | | Left | 1.00 (Ref.) | | | | | Right | 1.35 (0.71-2.58) | 0.36 | | | | Stone burdena, mm2 | 0.99 (0.99-1.00) | <0.001 | 1.00 (0.99-1.00) | 0.04 | | Tract lengtha | 1.01 (0.99-1.02) | 0.16 | | | | Renal pelvic obstruction | | None or mild | 1.00 (Ref.) | | | | | Moderate or severe | 0.83 (0.42-1.67) | 0.83 | | | | Number of calyxes involveda | 0.75 (0.56-1.00) | 0.05 | 1.36 (0.98-2.65) | 0.21 | | Stone density (HU)a | 0.99 (0.99-1.00) | 0.18 | | | | Prior treatment | | No | 1.00 (Ref.) | | | | | PNL | 1.28 (0.41-4.63) | 0.67 | | | | ESWL | 1.31 (0.08-NA) | 0.99 | | | | Endoscopic | 1.36 (0.24-NA) | 0.99 | | | | Multiple | 1.82 (0.52-54.36) | 0.33 | | | | Presence of staghorn | | No | 1.00 (Ref.) | | | | | Yes | 0.25 (0.12-0.48) | <0.001 | 0.30 (0.11-0.72) | 0.01 | | Number of stones | | Single | 1.00 (Ref.) | | | | | Multiple | 0.32 (0.14-0.66) | 0.03 | 0.42 (0.18-0.91) | 0.03 | | Guy’s stone scorea | 0.47 (0.33-0.67) | <0.001 | 0.53 (0.31-0.95) | 0.04 | | S.T.O.N.E. nephrolithometry scorea | 0.71 (0.57-0.86) | 0.001 | 0.95 (0.66-1.34) | 0.69 | | CROES scorea | 1.71 (1.24-2.37) | 0.001 | 0.89 (0.41-1.74) | 0.68 | | AUC of the model | | | 0.72 (0.70-0.76) | |
|
|
Univariate and multivariate binomial logistic regression analysis to assess predictors of stone-free status.
|
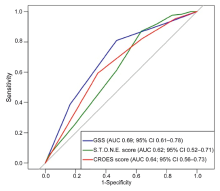
|
|
The receiver-operating curves for GSS, S.T.O.N.E. score and CROES score. GSS, the Guy’s stone score; S.T.O.N.E., the stone size, tract length, obstruction, number of involved calyces, and essence of stone; CROES, the Clinical Research Office of the Endourological Society; AUC, the area under the curve; CI, confidence interval.
|

| Parameter | GSS | S.T.O.N.E. nephrolithometry score | CROES score | | AUC (95% CI) | 0.69 (0.61-0.78) | 0.62 (0.52-0.71) | 0.64 (0.56-0.73) | | Accuracy (95% CI) | 0.75 (0.50-0.81) | 0.76 (0.61-0.81) | 0.61 (0.55-0.77) | | Best threshold | 2.5 | 7.5 | 202 | | Sensitivity (95% CI) | 0.81 (0.75-0.88) | 0.87 (0.62-0.98) | 0.59 (0.53-0.86) | | Specificity (95% CI) | 0.53 (0.41-0.68) | 0.37 (0.18-0.61) | 0.65 (0.31-0.78) | | NPV (95% CI) | 0.44 (0.29-0.55) | 0.45 (0.31-0.71) | 0.32 (0.27-0.47) | | PPV (95% CI) | 0.86 (0.83-0.93) | 0.83 (0.80-0.86) | 0.86 (0.80-0.91) |
|
|
GSS, S.T.O.N.E. nephrolithometry score and CROES score performances.
|
| [1] |
Turk C, Petrik A, Sarica K, Seitz C, Skolarikos A, Straub M, et al. EAU guidelines on interventional treatment for urolith-iasis. Eur Urol 2016; 69:475e82.
doi: 10.1016/j.eururo.2015.07.041
|
| [2] |
Assimos D, Krambeck A, Miller NL, Monga M, Murad MH, Nelson CP, et al. Surgical management of stones: American Urological Association/Endourological Society guideline, Part I. J Urol 2016; 196:1153e60.
doi: 10.1016/j.juro.2016.05.090
pmid: 27238616
|
| [3] |
Bryniarski P, Paradysz A, Zyczkowski M, Kupilas A, Nowakowski K, Bogacki R. A randomized controlled study to analyze the safety and ef?cacy of percutaneous nephrolithotripsy and retrograde intrarenal surgery in the management of renal stones more than 2 cm in diameter. J Endourol 2012; 26:52e7.
doi: 10.1089/end.2011.0235
pmid: 22003819
|
| [4] |
de la Rosette JJ, Opondo D, Daels FP, Giusti G, Serrano A, Kandasami SV, et al. Categorisation of complications and validation of the Clavien score for percutaneous neph-rolithotomy. Eur Urol 2012; 62:246e55.
doi: 10.1016/j.eururo.2012.03.055
pmid: 22487016
|
| [5] |
Armitage JN, Irving SO, Burgess NA; British Association of Urological Surgeons Section of Endourology. Percutaneous nephrolithotomy in the United Kingdom: results of a pro-spective data registry. Eur Urol 2012; 61:1188e93.
doi: 10.1016/j.eururo.2012.01.003
pmid: 22244778
|
| [6] |
Jackman SV, Docimo SG, Cadeddu JA, Bishoff JT, Kavoussi LR, Jarrett TW. The "mini-perc" technique: a less invasive alternative to percutaneous nephrolithotomy. World J Urol 1998; 16:371e4.
doi: 10.1007/s003450050083
pmid: 9870281
|
| [7] |
Lahme S, Zimmermanns V, Hochmuth A, Janitzki V. [Minimally invasive PCNL (mini-perc). Alternative treatment modality or replacement of conventional PCNL?]. Urologe 2008; 47:563e8.[Article in German].
doi: 10.1007/s00120-008-1708-3
|
| [8] |
Desai MR, Sharma R, Mishra S, Sabnis RB, Stief C, Bader M. Single-step percutaneous nephrolithotomy (microperc): the initial clinical report. J Urol 2011; 186:140e5.
|
| [9] |
Li X, He Z, Wu K, Li SK, Zeng G, Yuan J, et al. Chinese mini-mally invasive percutaneous nephrolithotomy: the Guangzhou experience. J Endourol 2009; 23:1693e7.
doi: 10.1089/end.2009.1537
|
| [10] |
Mishra S, Sharma R, Garg C, Kurien A, Sabnis R, Desai M. Prospective comparative study of miniperc and standard PNL for treatment of 1 to 2 cm size renal stone. BJU Int 2011; 108: 896e9.
doi: 10.1111/j.1464-410X.2010.09936.x
pmid: 21477212
|
| [11] |
Kamal W, Kallidonis P, Kyriazis I, Liatsikos E. Minituriazed percutaneous nephrolithotomy: what does it mean? Urolith-iasis 2016; 44:195e201.
|
| [12] |
Thomas K, Smith NC, Hegarty N, Glass JM. The Guy’s stone scoredgrading the complexity of percutaneous neph-rolithotomy procedures. Urology 2011; 78:277e81.
doi: 10.1016/j.urology.2010.12.026
pmid: 21333334
|
| [13] |
Okhunov Z, Friedlander JI, George AK, Duty BD, Moreira DM, Srinivasan AK, et al. S.T.O.N.E. nephrolithometry: novel sur-gical classi?cation system for kidney calculi. Urology 2013; 81: 1154e9.
doi: 10.1016/j.urology.2012.10.083
|
| [14] |
Opondo D, Gravas S, Joyce A, Pearle M, Matsuda T, Sun YH, et al. Standardization of patient outcomes reporting in percutaneous nephrolithotomy. J Endourol 2014; 28:767e74.
doi: 10.1089/end.2014.0057
pmid: 24571713
|
| [15] |
Al Adl AM, Mohey A, Abdel Aal A, Abu-Elnasr HAF, El Karamany T, Noureldin YA. Percutaneous nephrolithotomy outcomes based on S.T.O.N.E., GUY, CROES, and S-ReSC scoring systems: the ?rst prospective study. J Endourol 2020; 34:1223e8.
doi: 10.1089/end.2019.0856
|
| [16] |
Kumar S, Sreenivas J, Karthikeyan VS, Mallya A, Keshavamurthy R. Evaluation of CROES nephrolithometry nomogram as a preoperative predictive system for percutaneous nephrolithotomy outcomes. J Endourol 2016; 30:1079e83.
pmid: 27550775
|
| [17] |
Tailly TO, Okhunov Z, Nadeau BR, Huynh MJ, Labadie K, Akhavein A, et al. Multicenter external validation and com-parison of stone scoring systems in predicting outcomes after percutaneous nephrolithotomy. J Endourol 2016; 30:594e601.
doi: 10.1089/end.2015.0700
|
| [18] |
Zeng G, Wan S, Zhao Z, Zhu J, Tuerxun A, Song C, et al. Super-mini percutaneous nephrolithotomy (SMP): a new concept in technique and instrumentation. BJU Int 2016; 117:655e61.
doi: 10.1111/bju.13242
pmid: 26220396
|
| [19] |
Tiselius HG, Andersson A. Stone burden in an average Swedish population of stone formers requiring active stone removal: how can the stone size be estimated in the clinical routine? Eur Urol 2003; 43:275e81.
doi: 10.1016/S0302-2838(03)00006-X
|
| [20] |
Wu WJ, Okeke Z. Current clinical scoring systems of percu-taneous nephrolithotomy outcomes. Nat Rev Urol 2017; 14: 459e69.
doi: 10.1038/nrurol.2017.71
|
| [21] |
Mitropoulos D, Artibani W, Graefen M, Remzi M, Roupret M, Truss M, et al. Reporting and grading of complications after urologic surgical procedures: an ad hoc EAU guidelines panel assessment and recommendations. Eur Urol 2012; 61:341e9.
doi: 10.1016/j.eururo.2011.10.033
pmid: 22074761
|
| [22] |
Pietropaolo A, Proietti S, Geraghty R, Skolarikos A, Papatsoris A, Liatsikos E, et al. Trends of “urolithiasis: in-terventions, simulation, and laser technology” over the last 16 years (2000e2015) as published in the literature (PubMed): a systematic review from European section of Uro-technology (ESUT). World J Urol 2017; 35:1651e8.
doi: 10.1007/s00345-017-2055-z
|
| [23] |
Chung KJ, Kim JH, Min GE, Park HK, Li S, Del Giudice F, et al. Changing trends in the treatment of nephrolithiasis in the real world. J Endourol 2019; 33:248e53.
doi: 10.1089/end.2018.0667
pmid: 30628473
|
| [24] |
Ahmad AA, Alhunaidi O, Aziz M, Omar M, Al-Kandari AM, El-Nahas A, et al. Current trends in percutaneous nephrolithotomy: an internet-based survey. Ther Adv Urol 2017; 9:219e26.
doi: 10.1177/1756287217724726
|
| [25] |
Feng D, Hu X, Tang Y, Han P, Wei X. The ef?cacy and safety of miniaturized percutaneous nephrolithotomy versus standard percutaneous nephrolithotomy: a systematic review and meta-analysis of randomized controlled trials. Investig Clin Urol 2020; 61:115e26.
doi: 10.4111/icu.2020.61.2.115
|
| [1] |
Denis V. Krakhotkin,Volodymyr A. Chernylovskyi,Kemal Sarica,Arman Tsaturyan,Evangelos Liatsikos,Jurijus Makevicius,Nikolay Yu Iglovikov,Dmitry N. Pikhovkin. Diagnostic value ultrasound signs of stones less than or equal to 10 mm and clinico-radiological variants of ureteric colic[J]. Asian Journal of Urology, 2023, 10(1): 39-49. |
| [2] |
Leandro Blas,Masaki Shiota,Shohei Nagakawa,Shigehiro Tsukahara,Takashi Matsumoto,Ken Lee,Keisuke Monji,Eiji Kashiwagi,Junichi Inokuchi,Masatoshi Eto. Validation of user-friendly models predicting extracapsular extension in prostate cancer patients[J]. Asian Journal of Urology, 2023, 10(1): 81-88. |
| [3] |
Pankaj N. Maheshwari,Amandeep Arora,Mahesh S. Sane,Vivek Jadhao. Evaluating factors associated with the risk of hydrothorax following standard supracostal percutaneous nephrolithotomy[J]. Asian Journal of Urology, 2022, 9(3): 301-306. |
| [4] |
Abhay D. Mahajan,Lakshman P. Singh,Prashant P. Darakh,Sandeep T. Bathe,Martand G. Patil,Arpit R. Sharma. Uroflowmetric analysis and derivation of nomograms for normal paediatric Indian population between 5 to 15 years of age[J]. Asian Journal of Urology, 2022, 9(2): 125-131. |
| [5] |
Sujeet Poudyal. Current insights on haemorrhagic complications in percutaneous nephrolithotomy[J]. Asian Journal of Urology, 2022, 9(1): 81-93. |
| [6] |
Jiefeng Xiao,Shukai Zheng,Zhaolong Qiu,Kusheng Wu. Associations between IL-1RN variable number of tandem repeat, IL-1β (-511) and IL-1β (+3954) gene polymorphisms and urolithiasis in Uighur children of China[J]. Asian Journal of Urology, 2022, 9(1): 51-56. |
| [7] |
Abhishek Gajendra Singh,Sundaram Palaniappan,Shrikant Jai,Gopal Tak,Arvind Ganpule,Ravindra Sabnis,Mahesh Desai. The clinical outcomes of laser with suction device in mini-percutaneous nephrolithotomy[J]. Asian Journal of Urology, 2022, 9(1): 63-68. |
| [8] |
Dilip K. Mishra,Sonia Bhatt,Sundaram Palaniappan,Talamanchi V.K. Reddy,Vinothkumar Rajenthiran,Y.L. Sreeranga,Madhu S. Agrawal. Mini versus ultra-mini percutaneous nephrolithotomy in a paediatric population[J]. Asian Journal of Urology, 2022, 9(1): 75-80. |
| [9] |
Yiwei Wang, Liheng Gao, Mingxi Xu, Wenfeng Li, Yuanshen Mao, Fujun Wang, Lu Wang, Jun Da, Zhong Wang. A novel spherical-headed fascial dilator is feasible for second-stage ultrasound guided percutaneous nephrolithotomy: A pilot study[J]. Asian Journal of Urology, 2021, 8(4): 424-429. |
| [10] |
Braulio Omar Manzo,Jose David Cabrera,Esteban Emiliani,Hector Manuel Sánchez,Brian Howard Eisner,Jose Ernesto Torres. Impact of the adherence to medical treatment on the main urinary metabolic disorders in patients with kidney stones[J]. Asian Journal of Urology, 2021, 8(3): 275-279. |
| [11] |
Sarwar Noori Mahmood,Hewa Toffeq,Saman Fakhralddin. Sheathless and fluoroscopy-free retrograde intrarenal surgery: An attractive way of renal stone management in high-volume stone centers[J]. Asian Journal of Urology, 2020, 7(3): 309-317. |
| [12] |
Sudharsan Balaji,Arvind Ganpule,Thomas Herrmann,Ravindra Sabnis,Mahesh Desai. Contemporary role of multi-tract percutaneous nephrolithotomy in the treatment of complex renal calculi[J]. Asian Journal of Urology, 2020, 7(2): 102-109. |
| [13] |
Russell S. Terry,Glenn M. Preminger. Metabolic evaluation and medical management of staghorn calculi[J]. Asian Journal of Urology, 2020, 7(2): 122-129. |
| [14] |
Etienne Xavier Kellera,Vincent De Conincka,Steeve Doizia,Olivier Traxer. The role of ureteroscopy for treatment of staghorn calculi: A systematic review[J]. Asian Journal of Urology, 2020, 7(2): 110-115. |
| [15] |
Osman Ermis,Bhaskar Somani,Thomas Reeves,Selcuk Guven,Pilar Laguna Pes,Arun Chawla,Padmaraj Hegde,Jean de la Rosette. Definition, treatment and outcome of residual fragments in staghorn stones[J]. Asian Journal of Urology, 2020, 7(2): 116-121. |
|
|
|
|



